Which electron-dot diagram represents a molecule that has a polar covalent bond?
1). 2).
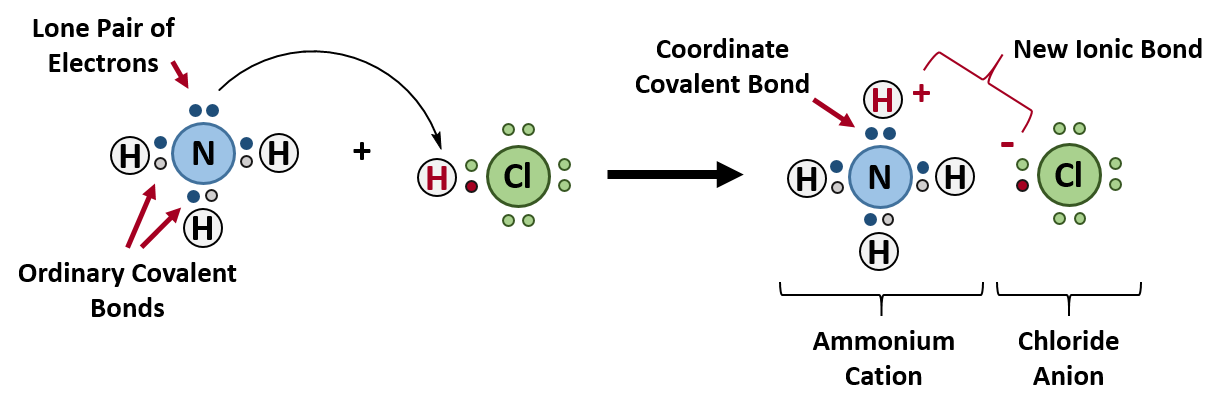
3). 4). ___ 2). A proton (H+) could form a coordinate covalent bond.

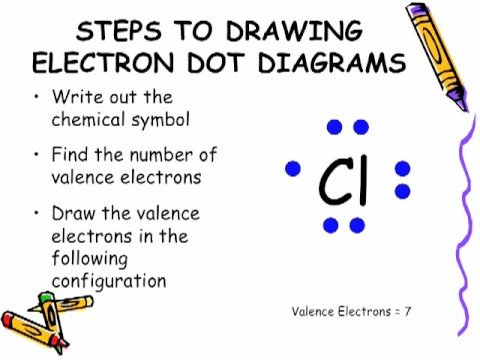
Draw a Lewis electron dot diagram for an atom or a monatomic ion. of how valence electrons interact, a simple way of representing those valence electrons .
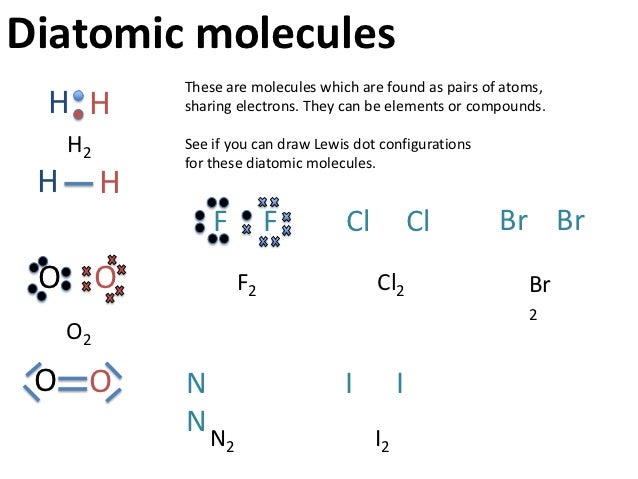
Electron Dot Structures – Helpful tools in thinking about bonding. Pictorial Electron dot structure – valence electrons are represented by dots placed around the.
Draw a Lewis electron dot diagram for an atom or a monatomic ion. of how valence electrons interact, a simple way of representing those valence electrons . Which electron-dot diagram represents a molecule that has a polar covalent bond?
1). 2).
3). 4).
___ 2). A proton (H+) could form a coordinate covalent bond.The diagram below represents a water molecule. 1)polar with polar covalent bonds 2)polar with nonpolar covalent bonds 3)nonpolar with polar covalent bonds 4)nonpolar with nonpolar covalent bonds This molecule is best described as 1)H2O 2)CCl4 3)NH3 4)H2 Which formula represents a nonpolar molecule containing polar covalent bonds?
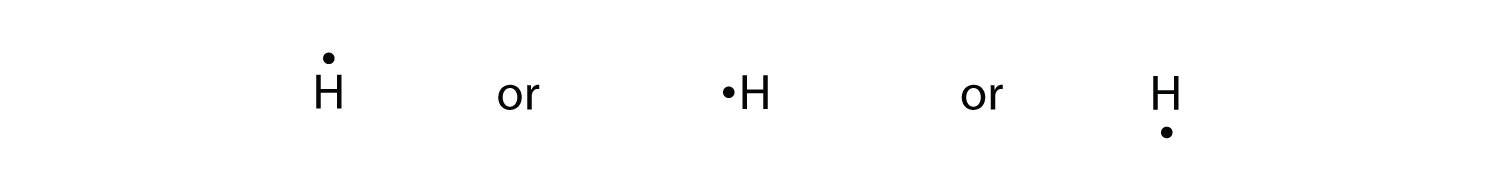
Lewis structures, also known as Lewis dot diagrams, Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDS), are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. Sep 24, · Lewis dot structures for a nonpolar molecule must show equal electron distribution throughout the molecule, thus having no dipole.
For a simple example: H2 is nonpolar. So H-H would be represented with H(two dots)H. 2.
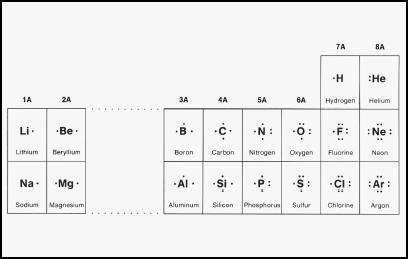
Fluorine has 7/8 valence electrons, therefore F is surrounded by 3 sets of pairs of dots and 1 single dot on one side. schematron.org: Resolved. Start studying Chem- Lewis structure.
Learn vocabulary, terms, and more with flashcards, games, and other study tools. Locate the element you are drawing an electron dot diagram for on the periodic table of elements.
Find the number of electrons in one atom of the element. The number of electrons is the same as the element’s atomic number.BBC – GCSE Bitesize: Dot-and-cross diagrams – elementsLewis structure – Wikipedia