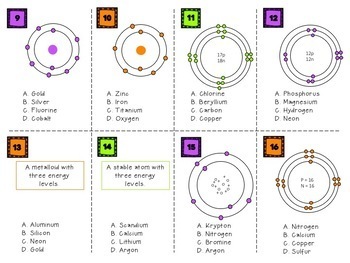
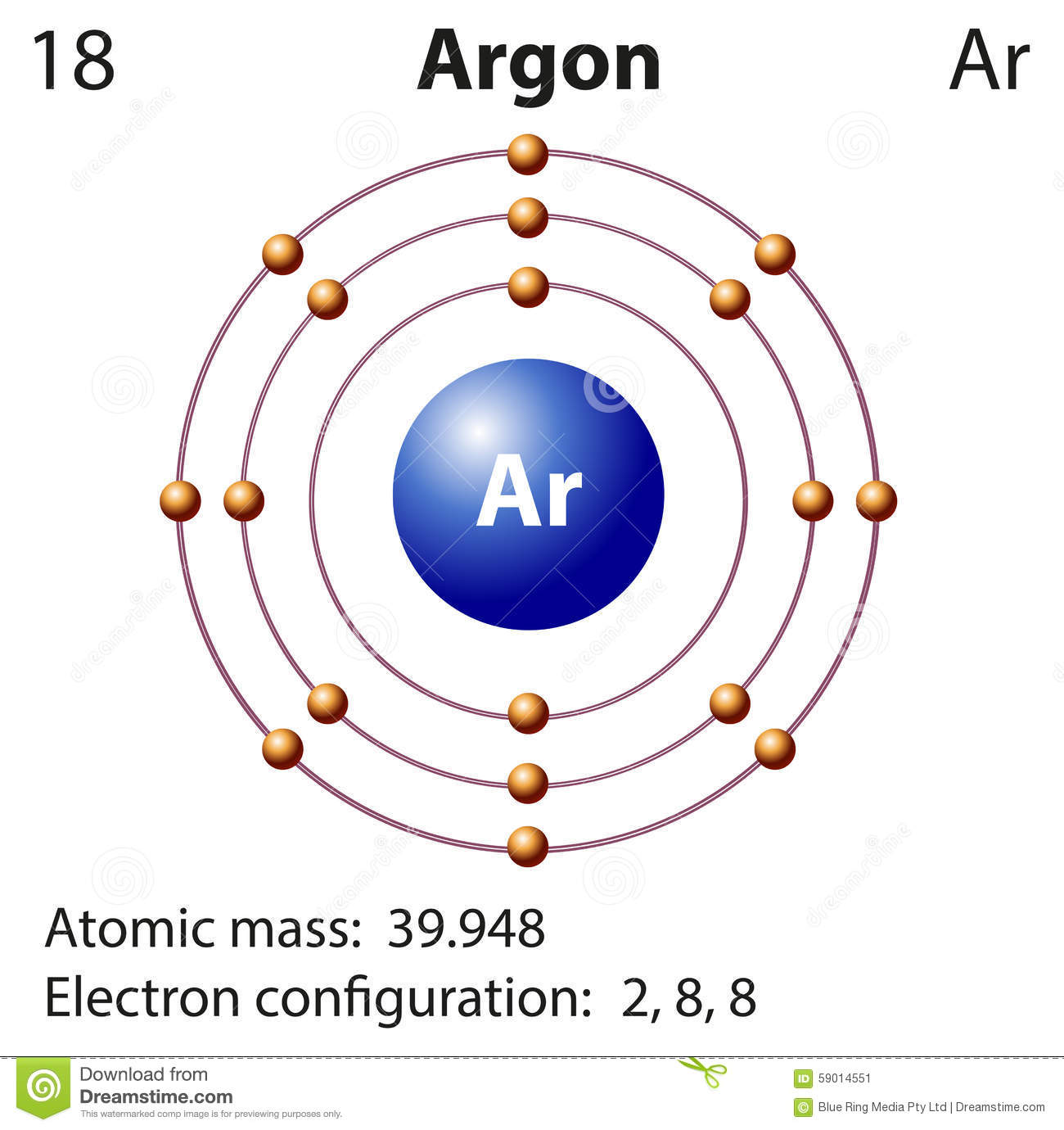
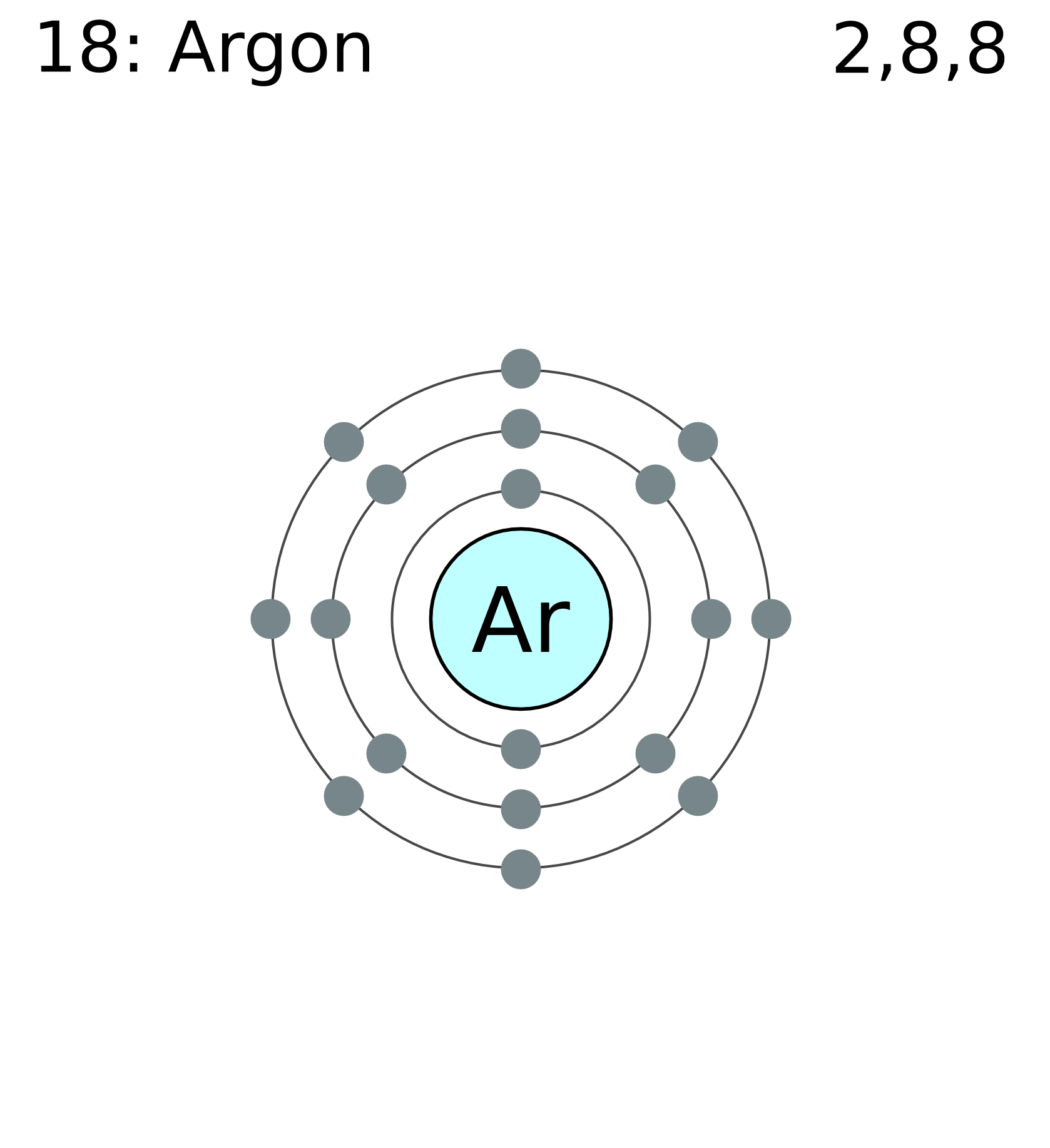
Here is a typical Bohr model, Draw a Bohr Model for an Argon atom. How many neutrons and protons does it have?
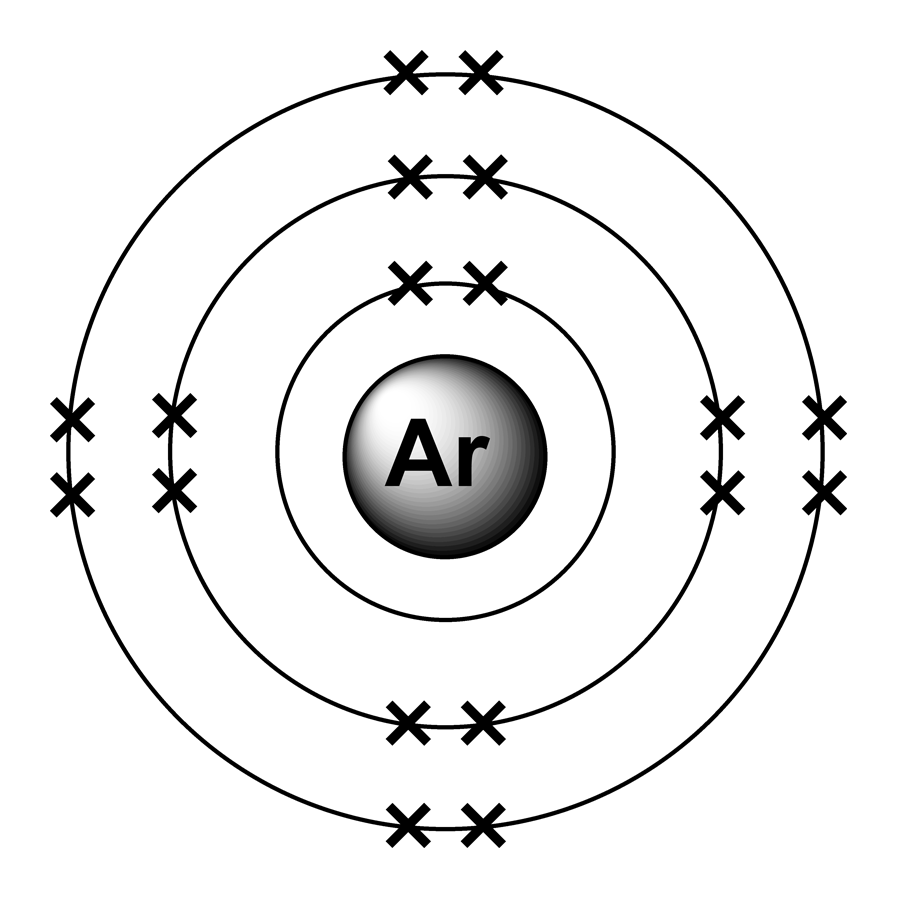
How many electrons does. Bohr model of noble gases, Neon, Argon, Krypton. determines all structures.
Bohr model Neon orbits and motion. de Broglie wave and periodic table.
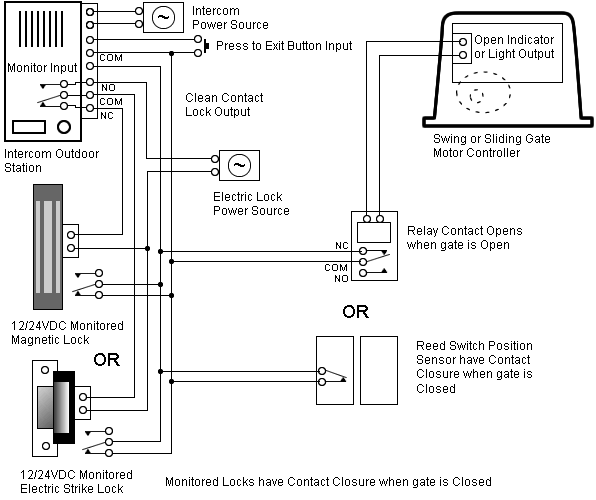
Last class, we determined that the Bohr Model is a planetary model in which the For example, there are 3 shells in the bohr diagram of Argon. Bohr Model Of Argon Atom Potassium Atom, Copper Atom, Atom Model Project, Bohr.
Visit chemical elements, crystals, melting points,[Bohr Model of Copper]. Last class, we determined that the Bohr Model is a planetary model in which the For example, there are 3 shells in the bohr diagram of Argon.In atomic physics, the Rutherford–Bohr model or Bohr model or Bohr diagram, presented by Niels Bohr and Ernest Rutherford in , a system consisting of a small, dense nucleus surrounded by revolving electrons —similar to the structure of the Solar System. “Bohr Model-In Niels Bohr proposed a model of the atom.
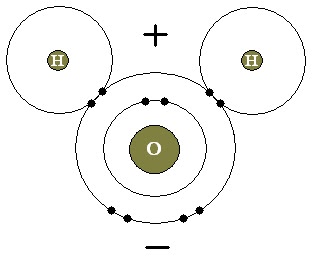
It is an atom where the nucleus is positively charged and is orbited by negatively charged electrons. This is a modification of the Rutherford model which was proposed earlier.
Facts Date of Discovery: Discoverer: Sir William Ramsay Name Origin: From the Greek word argon (inactive) Uses: Lighting Obtained From: air Related Links None available MLA Format for Citing This Page Bentor, Yinon. Chemical schematron.org – Argon. Dec 30, · Argon (Ar) atom Bohr model video using the Periodic Table to determine the number of Protons, neutrons, the Atomic Mass, Atomic Number, number .
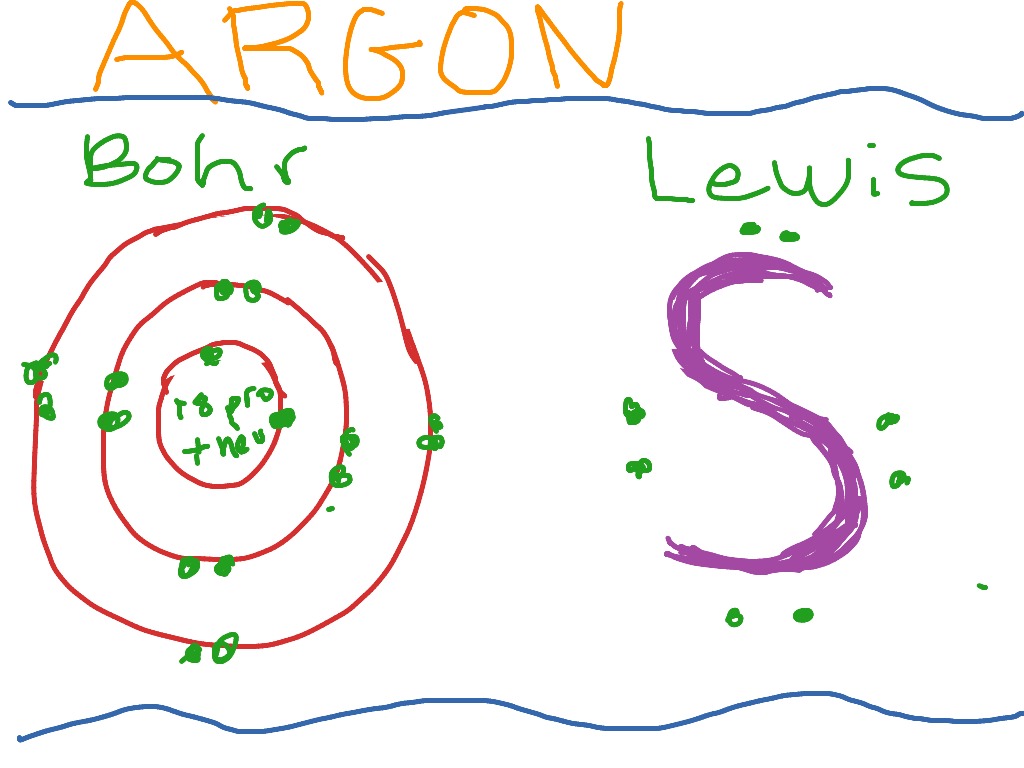
A Bohr diagram is a simplified visual representation of an atom that was developed by Danish physicist Niels Bohr in The diagram depicts the atom as a positively charged nucleus surrounded by electrons that travel in circular orbits about the nucleus in discrete energy levels.File argon (Ar) Bohr schematron.org – Wikimedia CommonsBohr model – Wikipedia