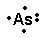
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. To facilitate our understanding of how valence electrons interact. These diagrams show the symbol of the element with as many dots around it as there are electrons in the outermost energy level.
I’d like you to get some practice by drawing electron dot diagrams for these elements now Arsenic has five. In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. To facilitate our understanding of how valence electrons interact.
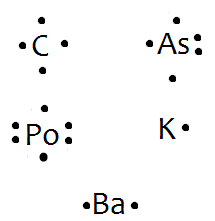
Arsenic. Se. Selenium.
Br. Bromine.
Kr . Chemists use these diagrams to visualize the association of valence electrons To draw a Lewis dot structure for an atom, you must know how many.
The left diagram shows a Lewis dot structure of sodium with. Likewise, they ..
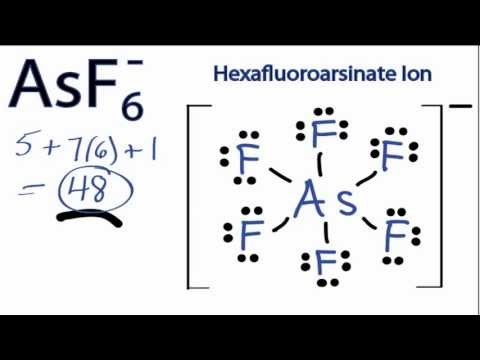
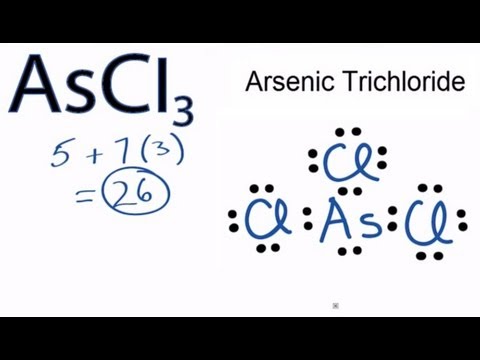
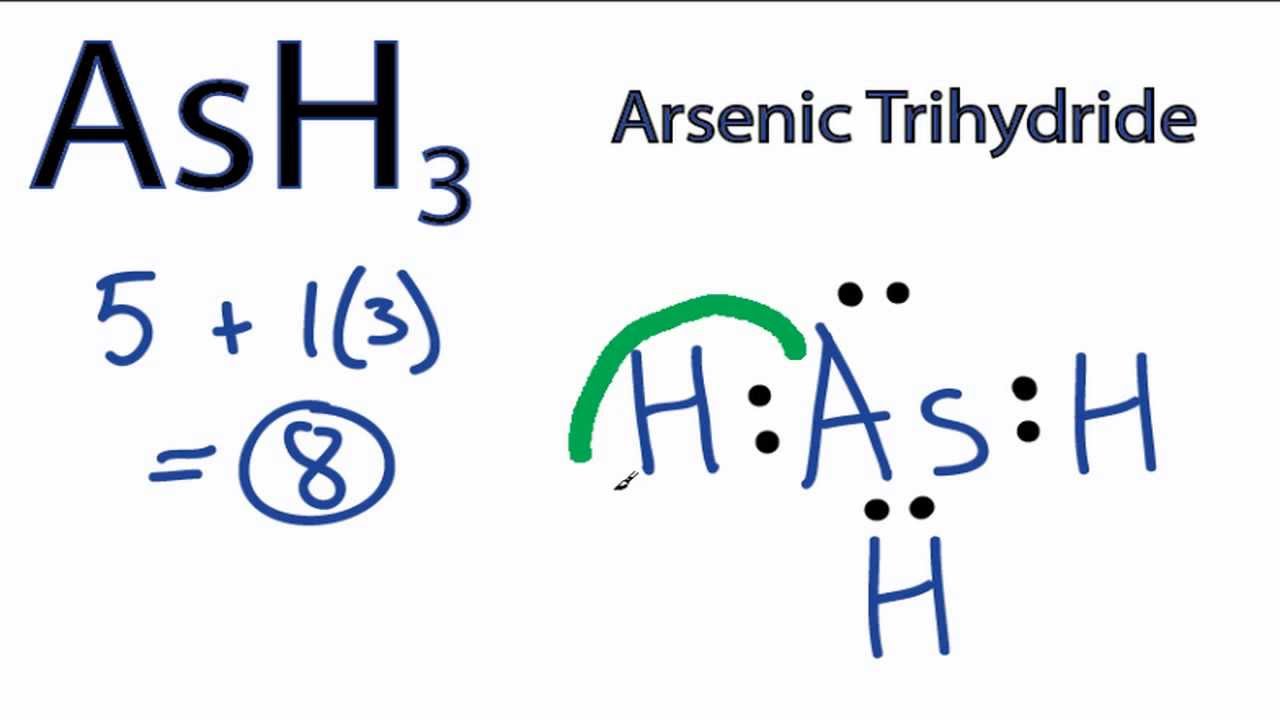
A Lewis structure shows an arsenic atom single bonded to three fluorine atoms.Periodic Table of Elements Element Arsenic – As. Comprehensive data on the chemical element Arsenic is provided on this page; including scores of properties, element names in many languages, most known nuclides of Arsenic.
There are two types of diagrams one is the Lewis diagram the other is the Electron dot diagram. To make the electron dot diagram you put the electron symbol and put a dot on o ne of the sides for each period (you don’t count the middle section.)Ne: = Neon dot diagram ‘ ‘. The answer would be Arsenic (As).
An electron-dot diagram is a graphical representation of the valence electrons of a certain element. The chemical symbol of an element placed in the middle and the valence electrons are represented by dots/5(7). Arsenic (As) has an atomic mass of Find out about its chemical and physical properties, states, energy, electrons, oxidation and more. Because Arsenic is in group 15 it will receive 5 valence electrons Start out by adding one dot around the symbol for Arsenic.
You’ll end up with 4 around it and all you have to do is add another one.What is the correct Lewis structure for arsenic? | SocraticLewis Diagrams for Atoms