13+. 8+.
Electrons that are located in the outermost energy level. Krypton. Lewis (Dot) Diagram: Even atoms with more than. 20 electrons are easy.

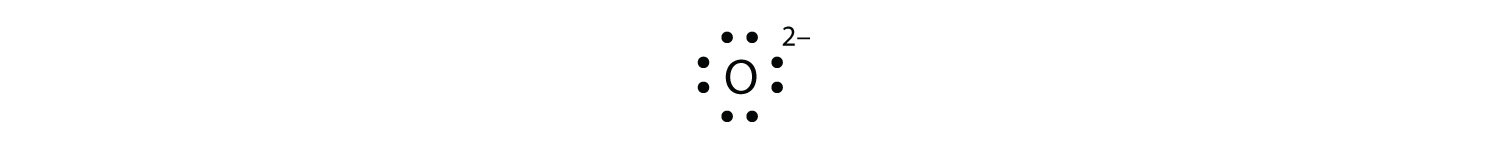
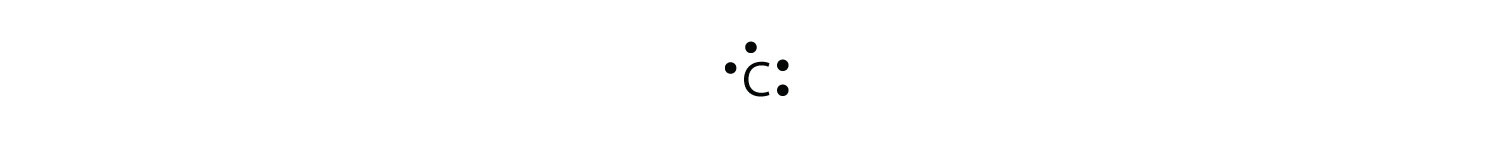
The electron-dot structure can be used for any elements. It just shows how electrons are distributed between bonds or number of valence. The electron-dot structure can be used for any elements. It just shows how electrons are distributed between bonds or number of valence.
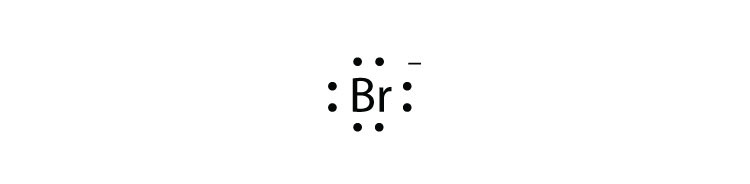
To answer your question, you would draw the resonance structure of each Lewis Dot Structure and then apply Formal Charge calculation to. To answer your question, you would draw the resonance structure of each Lewis Dot Structure and then apply Formal Charge calculation to.Krypton difluoride, or KrF 2, has the Lewis structure of a krypton atom with 3 lone pairs, single bonded to two fluorine atoms, each also containing 3 lone pairs. Krypton has 8 valence electrons, whereas fluorine contains 7 valence electrons.
A lone pair is a valence electron pair not shared with other atoms. Krypton (Kr) has an atomic mass of Find out about its chemical and physical properties, states, energy, electrons, oxidation and more.
Electron Distributions Into Shells for the First Three Periods. A chemical element is identified by the number of protons in its nucleus, and it must collect an equal number of electrons if .
Nov 07, · Best Answer: The electron-dot structure can be used for any elements. It just shows how electrons are distributed between bonds or number of valence electrons.
The dot-structure is drawn by laying out the element symbol and putting dots (resembling valence electrons) around schematron.org: Resolved. Krypton has a full outer shell of eight electrons, so it would be eight dots, either around the outer circle if you’re doing a full circle diagram, or just two dots above, below, to the left, and to the right of the Kr.What is the electron dot diagram for kryptonPeriodic Table of Elements: Krypton – Kr (schematron.org)