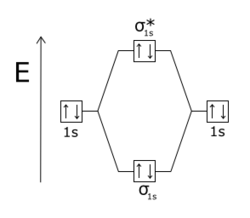
Atomic orbitals available for making Molecular Orbitals are 1s from each Helium. And total Since the bond order is zero so that He2 molecule does not exist. Atomic orbitals available for making Molecular Orbitals are 1s from each Helium.
And total Since the bond order is zero so that He2 molecule does not exist. This molecular orbital treatment can explain why H2 exists but He2 does not.
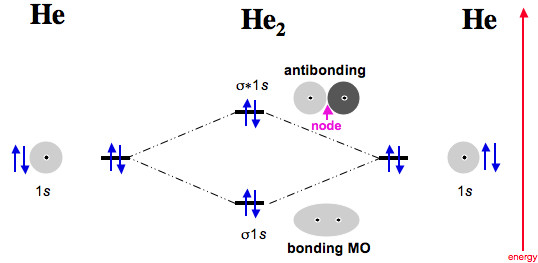
Draw a complete MO diagram for all the bonds in ethene. In He2 (dihelium), the two 1s atomic orbitals overlap to create two molecular orbitals: sigma(1s) and sigma(1s)*.
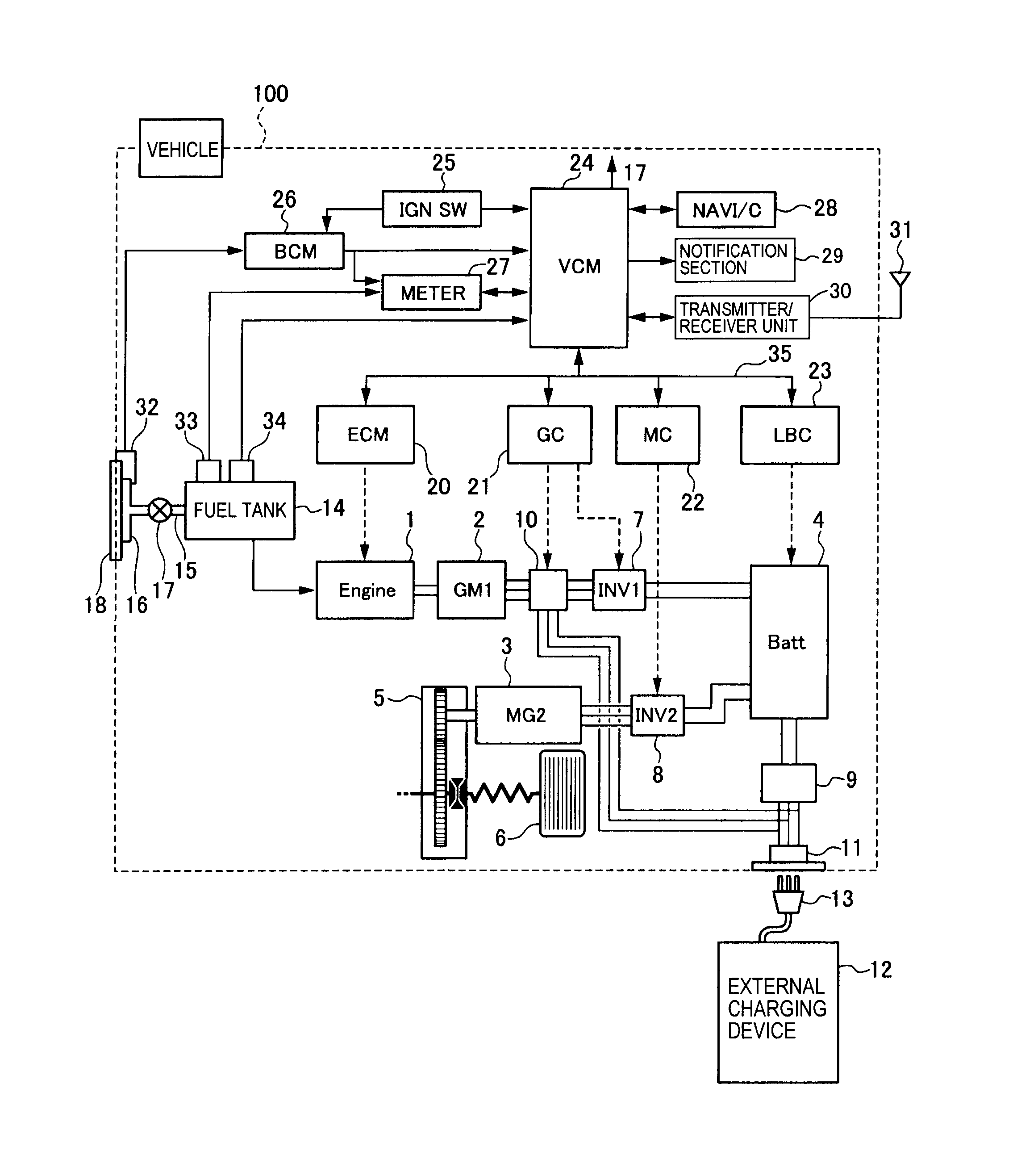
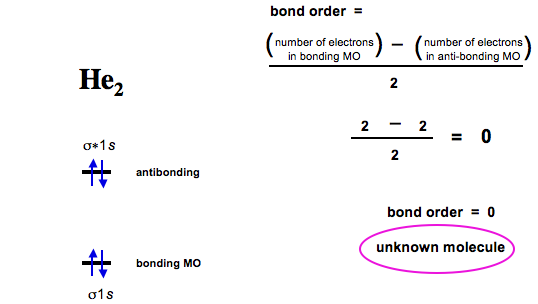
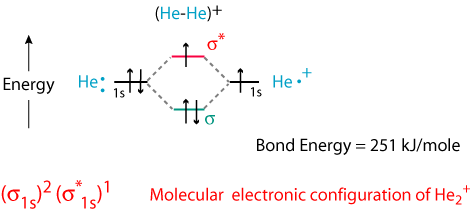
You fill these molecular orbitals. A molecular orbital explicitly describes the spatial distribution of a single Energy Level Diagrams He2 has bond order 0 [(2 − 2)/2 = 0], and we can make H+.The energy-level diagram for He2 is shown above, the two electrons in each of the 1s atomic orbital give total of 4 electrons in this molecule.
Two are placed in the bonding orbital, the other two in antibonding orbital. The bond order = 1/2 x (Number of Bonding Electrons – Number of Antibonding Electrons) = .
The molecular orbital energy-level diagram, which is a diagram that shows the relative energies of molecular orbitals, for the H 2 molecule is shown in Figure On either side of the central ladder are shown the energies of the 1 s orbitals of atoms A and B, and the central two-rung ladder shows the energies of the bonding and antibonding. The orbital correlation diagram in predicts the same thing–two electrons fill a single bonding molecular orbital. To further demonstrate the consistency of the Lewis structures with M.O.
theory, we will formalize a definition of bond order–the number of bonds between atoms in a molecule. Molecular Orbital Diagrams of Diatomic Molecules Introduction: In chemistry molecular orbital (MO) theory is a method for determining molecular structure in which electrons are not assigned to individual bonds between atoms, but are treated as moving under the influence of the nuclei in the whole molecule. The reason oh He2 Molecule to not exist can be explained on the basis of 1)MOLECULAR ORBITAL THEORY.
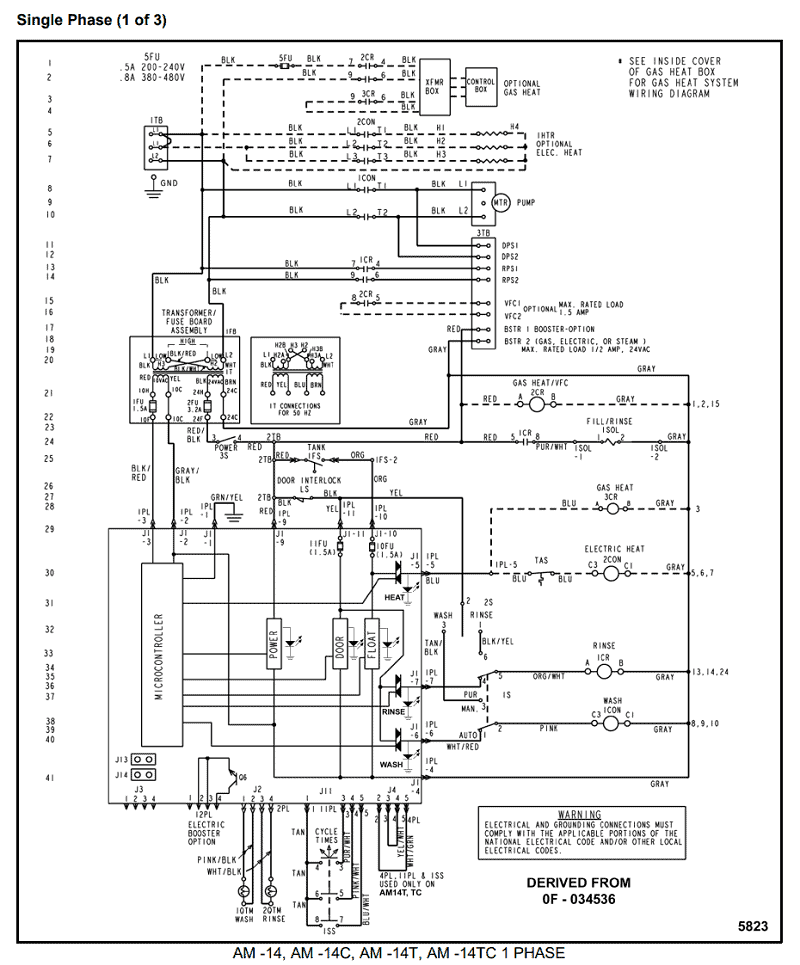
He has configuration of 1s2, if we draw its MOT DIAGRAM, 2 e’s enter the Bonding molecular Orbital and 2 e’s enter the AntiBonding molecular Orbital, thus net effect of the anti bonding and bonding is cancelled.Chemical bonding – Molecular orbitals of H2 and He2 | schematron.orgSparkNotes: Molecular Orbitals: Molecular Orbital Theory