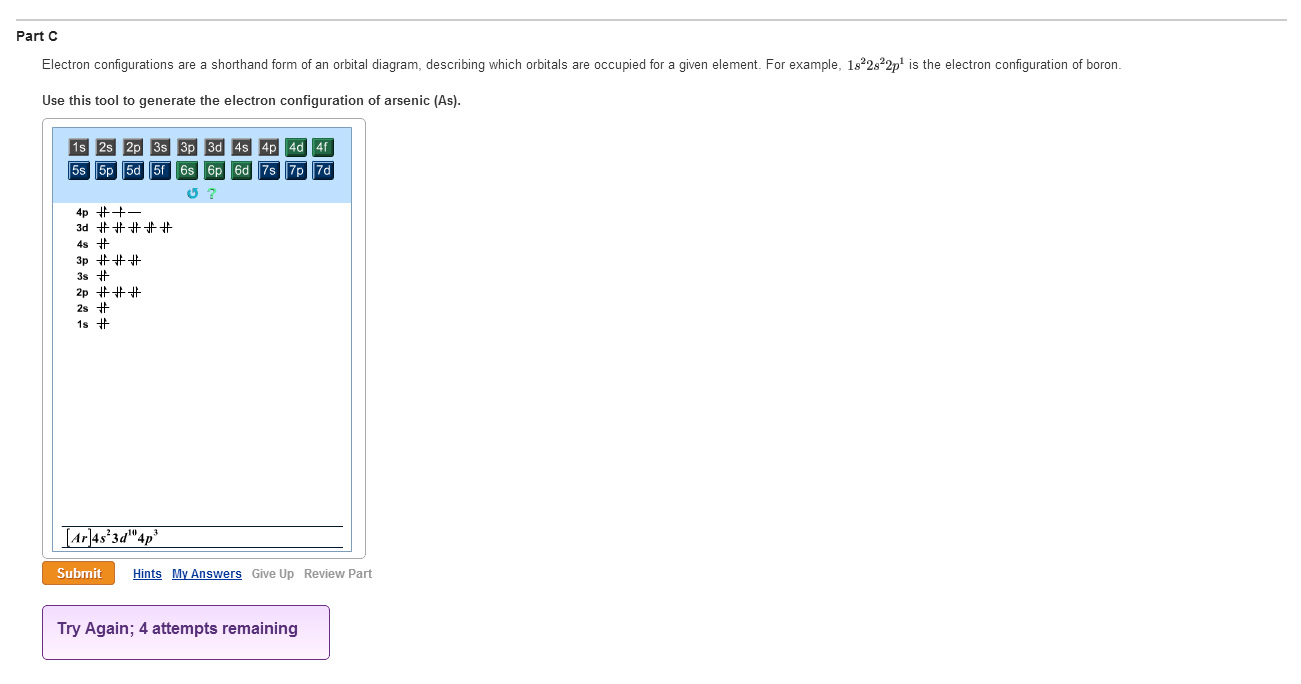
Because the 4p section has 3 orbitals, but Arsenic ends with 4p3.
It’ll want to leave as few orbitals empty, so you have three arrows pointing up. The orbital diagram of arsenic can be written as 1s2 2s2 2p6 3s23p6 4s2 3d10 4p3.

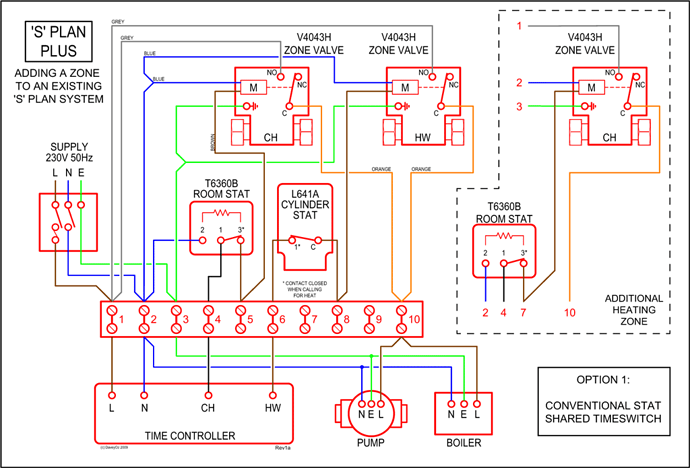
Arsenic has 33 electrons, including 3 in itsoutermost shell. schematron.org!
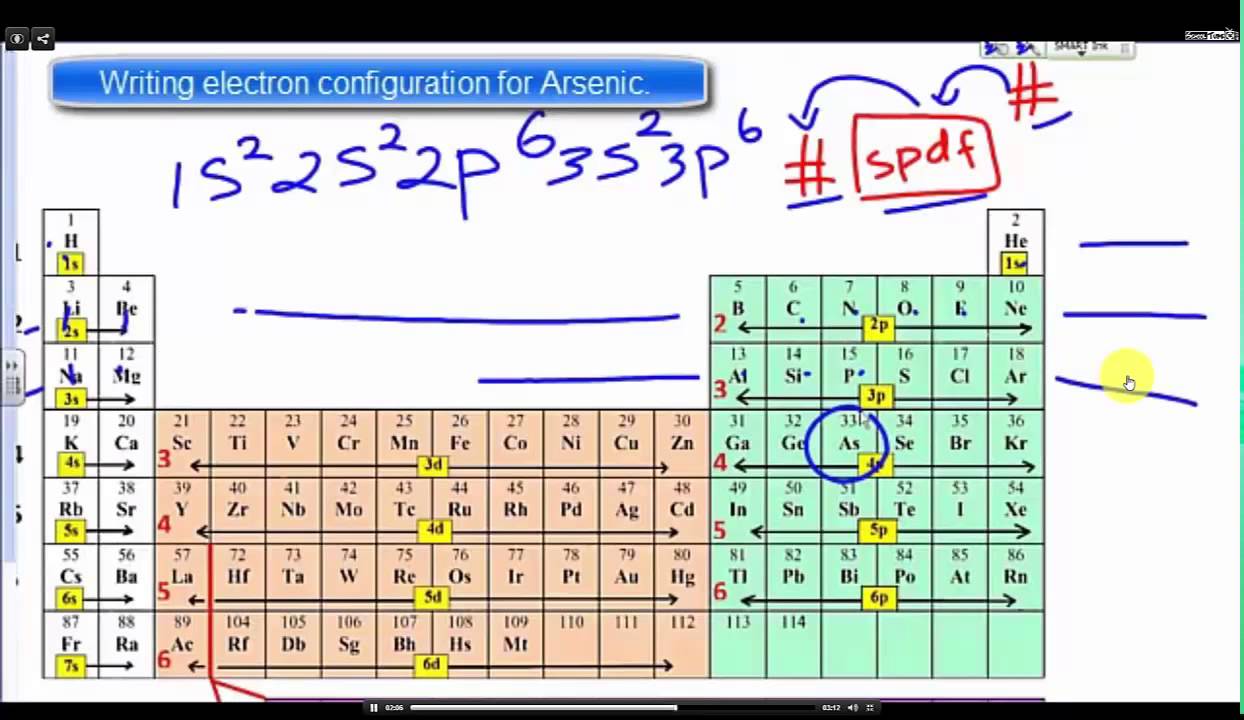
Arsenic atomic orbital and chemical bonding information. There are also tutorials on the first thirty-six elements of the periodic table.
Find an answer to your question Complete the orbital diagram for arsenic. Oxidation States, ±3,+5. Electrons Per Shell, 2 8 18 5.
Electron Configuration, [ Ar] 3d10 4s2 4p3. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p3.
Orbital Diagram. 1s.
↿⇂.Get thought from our ideas library or Orbital diagram for arsenic from Diagram gallery and create your home layout. Through the thousand photographs on the web about Orbital diagram for arsenic, selections the best series along side largest quality only for you all, and Gallery for > orbital diagram arsenic is in truth among images libraries inside our best possible footage gallery.

Nickel is in the. 4th.
energy level, d. block, 7th.
column, this means that the electron configuration will end. 3d8.
with the. d.
orbital being one level lower than the energy level it is on. orbital diagram for arsenic see more ideas approximately from diagram electron configuration gallery & create your house design images related to pictures as well help you in locating the solution are seeking about itOrbital diagram for arsenic.
Arsenic (As) has an atomic mass of Find out about its chemical and physical properties, states, energy, electrons, oxidation and more. The orbital notation of arsenic (As) is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3See the Related Questions for the electron configuration of all the elements.What is the Orbital Filling Diagram for Arsenic? | Yahoo AnswersWhat is the Orbital Filling Diagram for Arsenic?
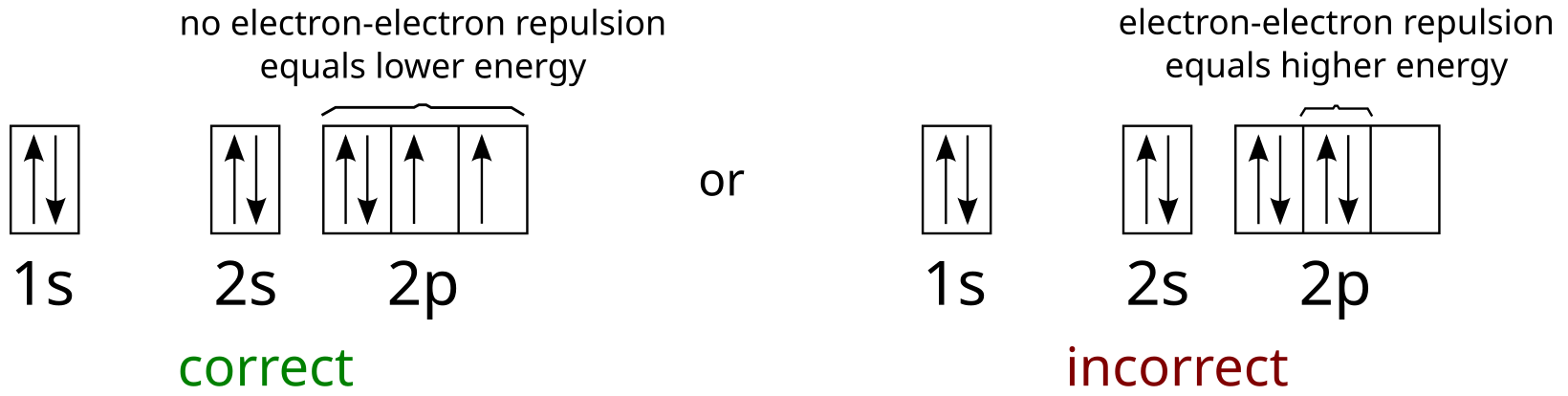
| Yahoo Answers