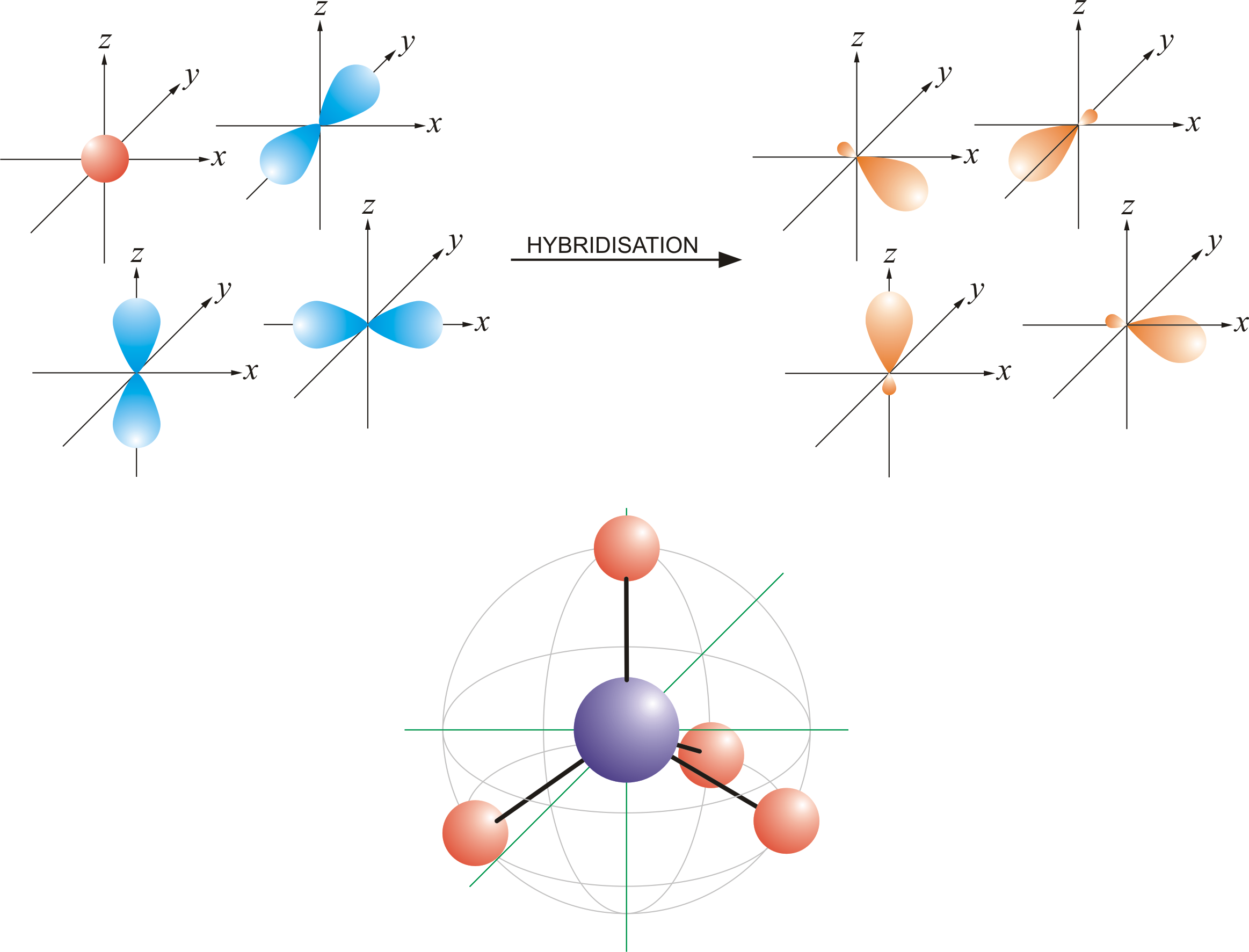
So no, the atom doesn’t have to get excited to 1s2 2s1 2p3 before In the case of sp3 hybridization, say in methane, the carbon s orbital. Orbital hybridization is essentially a process of mixing orbitals together and spitting out new ones that are all identical in “symmetry” and.
The atomic number of carbon is 6, which is also the number of The orbital diagram shows how the electrons are arranged within each sublevel. rule, each orbital must contain one electron each with the same spin, before.

The bonds in a methane (CH4) molecule are formed by four separate but equivalent orbitals; a single 2s and three 2p orbitals of the carbon hybridize into four. Drawing out the orbital diagram for a given molecule including the hybridized lobes.* Just like in methane molecule, each carbon atom undergoes sp 3 hybridization in the excited state to give four sp 3 hybrid orbitals in tetrahedral geometry.
* The two carbon atoms form a σ sp 3-sp 3 bond with each other due to overlapping of sp 3 hybrid orbitals along the inter-nuclear axis. sp 3 Hybridization.
The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. The p orbitals are equal in energy and said to be degenerate. The two singly occupied p orbitals can be utilized for bonding to give methylene CH 2, an unstable free radical (Figure 3).
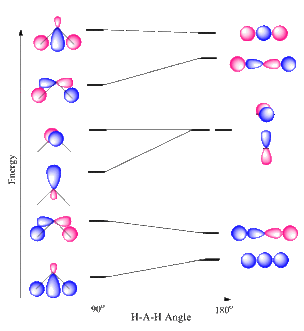
Because this type of sp hybridization only uses one of the p orbitals, there are still two p orbitals left which the carbon can use. Those p orbitals are at right angles .
What does the atomic orbital diagram of carbon look like before #sp^3# hybridization?
Orbital hybridization is essentially a process of mixing orbitals together and spitting out new ones that are all identical in “symmetry” and “composition” to the orbital(s) from the other, incoming atom(s). Solution: Consider the electron configuration of a carbon schematron.org the orbital diagram of carbon before sp3 hybridization.
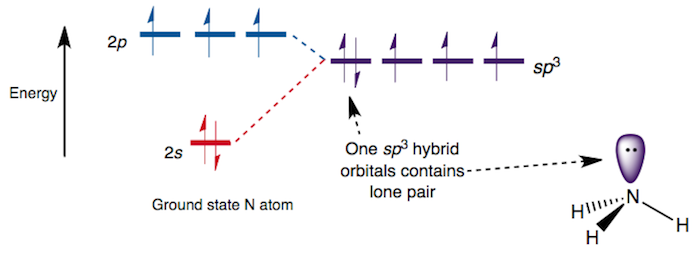
Problem. Consider the electron configuration of a carbon atom.
Write the orbital diagram of carbon before sp 3 hybridization. Next.
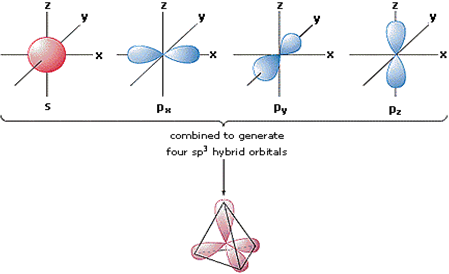
Practice Problems.What does the atomic orbital diagram of carbon look like before sp^3 hybridization? | SocraticConsider the electron configuration of a carbon atom.?
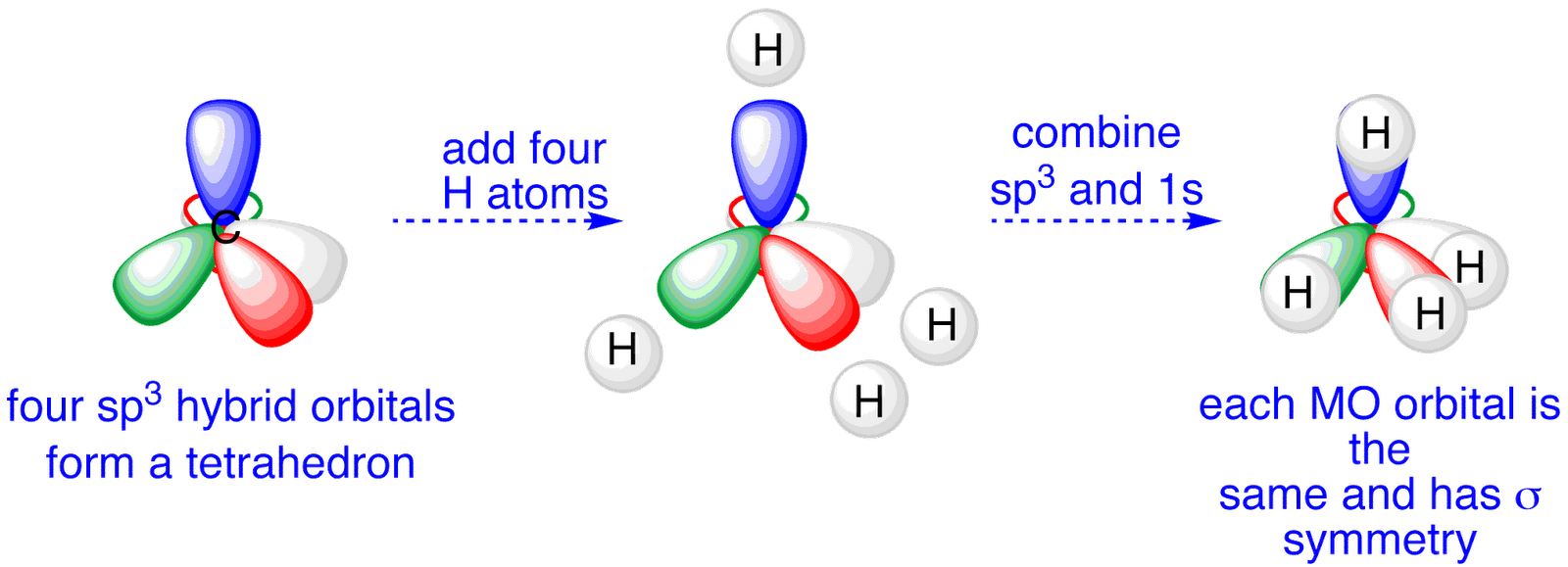
| Yahoo Answers