Since the 4s orbital is higher in energy, its electrons will be removed first. Not that it matters here, though, because exactly 5 electrons are.
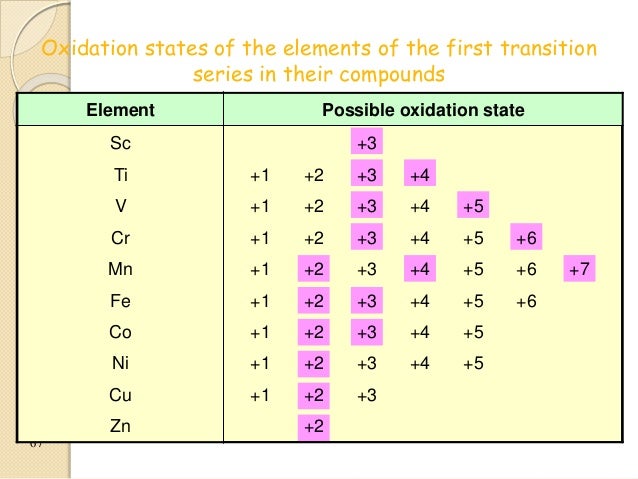
can be accommodated in the metal d orbitals. • d0 ions – Ti4+, Zr4+, V5+, Ta5+, Cr6+, Mo6+, etc. • d1 ions .
σ-ML4 Tetrahedral MO Diagram e. Answer to Write orbital diagrams for each of these ions.
V5+,Cr3+,Ni2+,Fe3+ Determine if the ion is diamagnetic or paramagnetic. V. (1) Question: What is the maximum number of electrons each orbital (s, p, d, f) can transition metals with their common oxidation states: A) V2+. B) V3+. C) V5+.
This is what I believe to be the orbital diagram of vanadium. I’m not certain.
If I’m wrong – please let me know if I am. Best wishes, Charlie Grame.So let’s look at the Auf Bau diagram which actually show this for us, okay so down here we have the 1s orbital but the 1 dash indicates that there’s 1 orbital within the 1s sublevel which makes sense that it is the lowest in energy, it’s its first principle energy level.
Write orbital diagrams for each of these ions. A.

V^5+ B. Cr^3+ C.
Ni^2+ D. Fe^3+ E. Determine if the following ions are diamagnetic or paramagnetic.
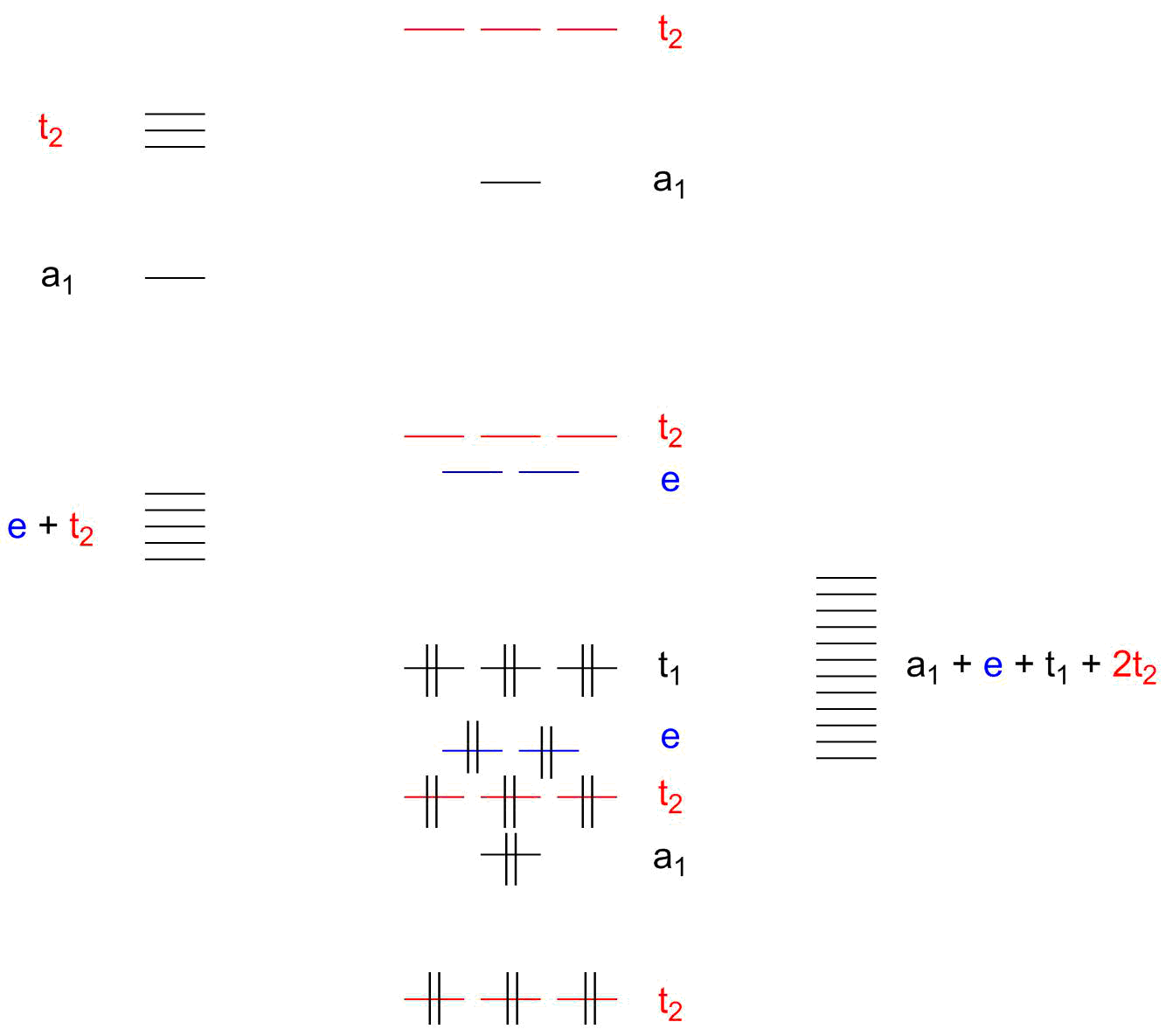
A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) molecular orbital method in particular. An orbital diagram naturally leads to the writing of an electron configuration. The electron configuration for chromium is: 1s22s22p63s23p64s23d4 The orbital diagram above is formatted in such a manner as to place the various orbital types at different energy levels.
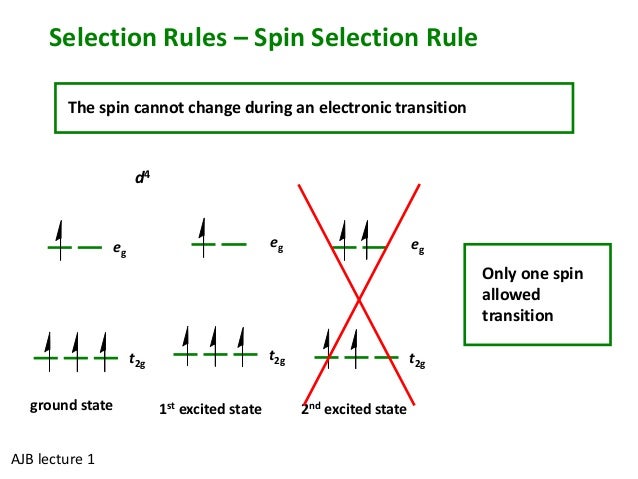
Nov 06, · This video shows you how to write the electron configuration for the vanadium atom and many of its ions – V, V2+, V3+, and V5+.Write orbital diagrams for each of these ions.? | Yahoo AnswersMolecular orbital diagram – Wikipedia