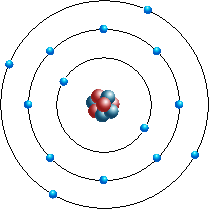
So for the element of MAGNESIUM, you already know that the atomic number tells you the number of electrons. That means there are 12 electrons in a. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun.
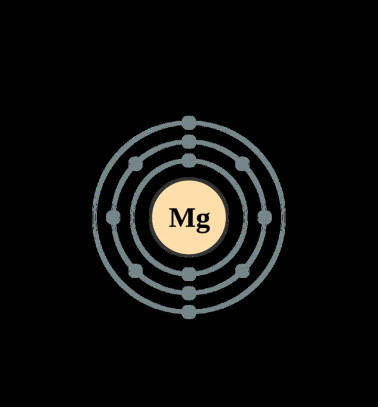
In the Bohr model, electrons are. Magnesium, Mg, has 12 electrons distributed as: 1st shell 2 electrons, 2nd shell 8 electrons and third shell 2 electrons. See how to draw here.
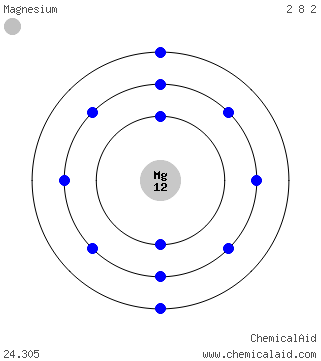
Magnesium at Chemical schematron.org Basic Information Number of Protons/ Electrons: Number of Neutrons: 12 [Bohr Model of Magnesium], Number of . Magnesium, Mg, has 12 electrons distributed as: 1st shell 2 electrons, 2nd shell 8 electrons and third shell 2 electrons.
See how to draw here.The bohr Rutherford diagram for oxygen has 8 protons and 8 neutrons. There are 2 electrons on the first orbital and six on the second. The bohr Rutherford diagram for oxygen h as 8 protons and.
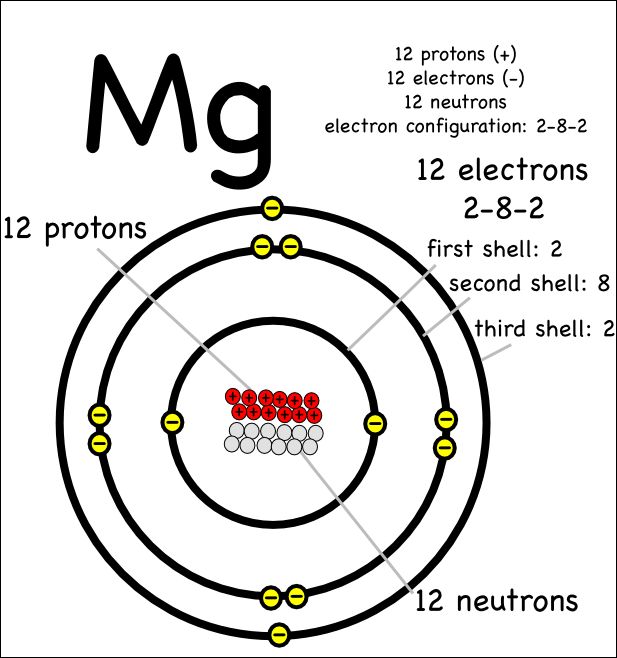
Feb 27, · I have to draw a Bohr Diagram for Magnesium and i ave to show how it forms a stable ion. What exactly does that mean schematron.org: Resolved. Bohr Model for Magnesium by Jackie Moore – October 15, Bohr diagram for magnesium satisfying the octet rule, this is the answer to the bohr electron configuration drawing of magnesium after it has completed the octet rule in the table talk question of lesson Magnesium has 12 protons and 12 electrons. The first electron shell of a Bohr model holds 2 electrons.
The second holds 8. So far, 10 of magnesium’s 12 electrons have been used, so only 2 remain.
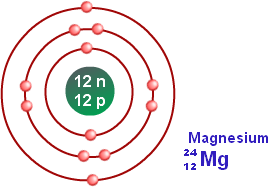
The remaining 2 are placed in the third electron shell, which is full when it holds 8 electrons.schematron.org: Magnesium: Orbital and Bonding InfoWhat would a Bohr Model for magnesium look like? | Socratic