Electron Configurations and Orbital Diagrams KEY. Draw orbital diagrams for the following elements: 1.
phosphorus. ↑↓.
↑↓ 4. germanium. ↑↓.
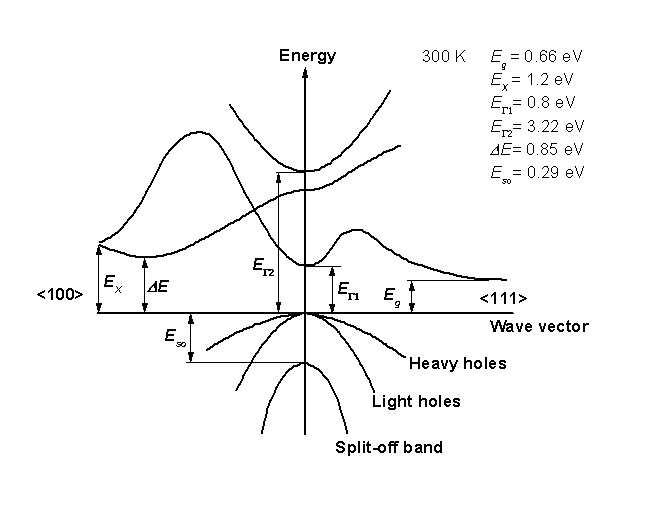
↑↓. Mendeleev’s Predicted Properties of Germanium (“eka.
Silicon”) and Its Use the orbital diagram to find the third and eighth electrons. PROBLEM: Write a set of . schematron.org!

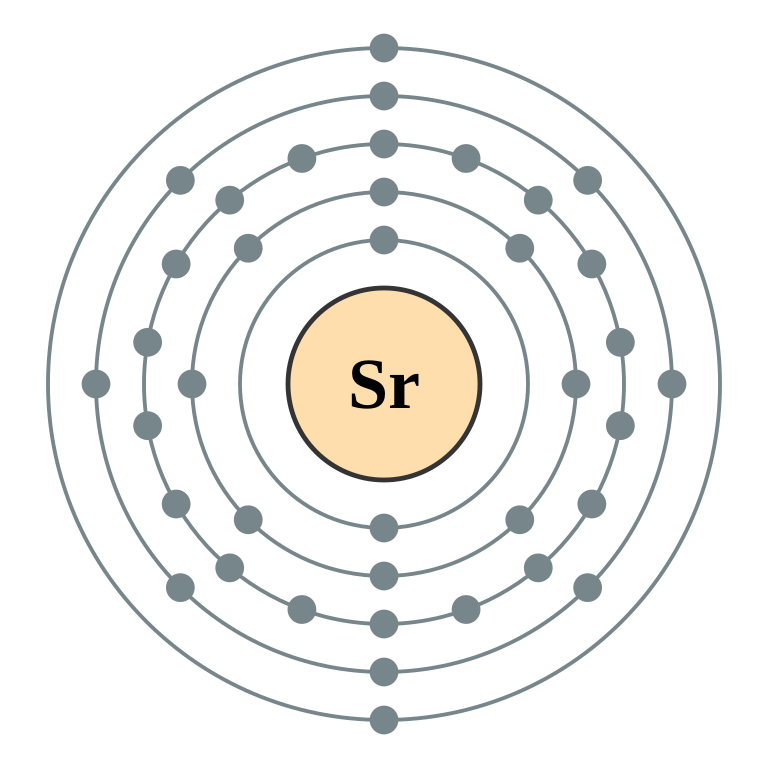
Germanium atomic orbital and chemical bonding information. There are also tutorials on the first thirty-six elements of the periodic table.
Oxidation States, +4,2. Electrons Per Shell, 2 8 18 4. Electron Configuration, [Ar] 3d10 4s2 4p2.
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p2. Orbital Diagram.
1s. ↿⇂. 2s.
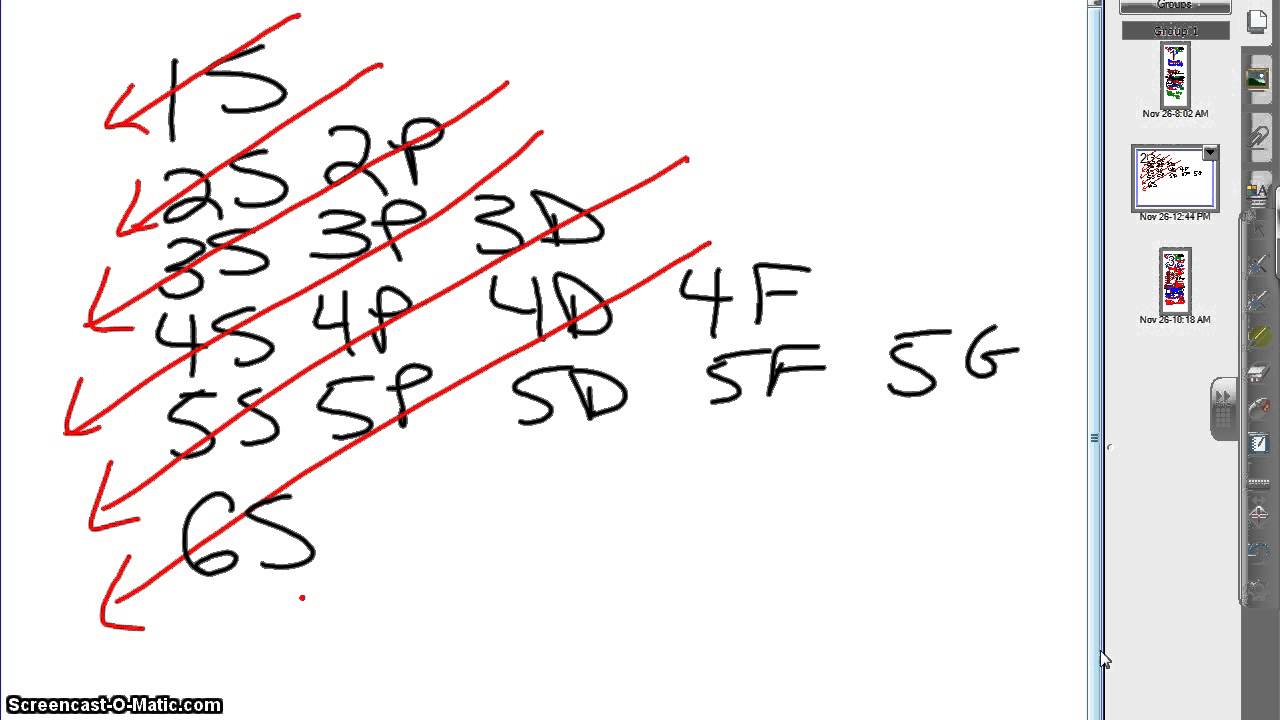
Use it to learn, and these orbital diagrams won’t be a problem! how do you draw the orbital diagram for an atom of germanium (Z = 32)?.Start studying Quantum Mechanics #3 Bohr Model, Electron Config, and Orbital Diagrams.
Learn vocabulary, terms, and more with flashcards, games, and other study tools. A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) molecular orbital method in particular.
Orbital Notation is a way to show how many electrons are in anorbital for a given element. They can either be shown with arrowsor circles. One arrow represents one electron in a shell. Twoarrows will be pointing differently; one up and one down to show amaximum of two electrons with different spin.
Germanium (Ge) has an atomic mass of Find out about its chemical and physical properties, states, energy, electrons, oxidation and more. Comprehensive information for the element Germanium – Ge is provided by this page including scores of properties, element names in many languages, most known nuclides and .Electron configuration – UBC WikiWhat is the quantum orbital notation of germanium