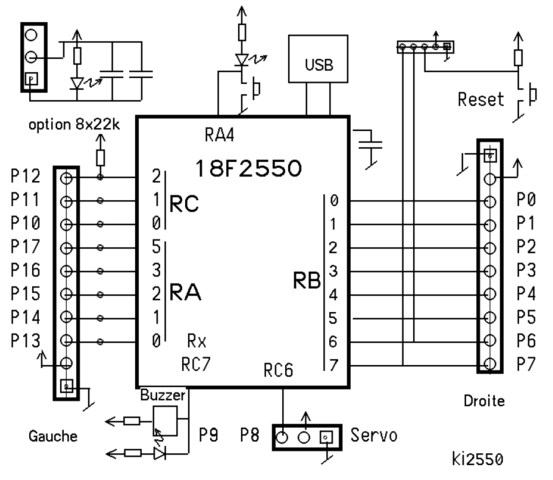
It is recommended to name the SVG file “Ammonia MO schematron.org” – then the template Vector Introduction to Inorganic Chemistry/Molecular Orbital Theory. Answer to For a molecular orbital diagram of ammonia (NH3), where the nitrogen atom is sp3 hybridized, the lone pair of electrons.
Drawing and calculating molecular orbitals of ammonia. MO diagram in Figure – a1 symmetry orbitals • Bondings, nonbonding, and antibonding MO – e.
the polyatomic molecules H2O, NH3, BH3 and SiH4 using group theory is reported. . Step 4.
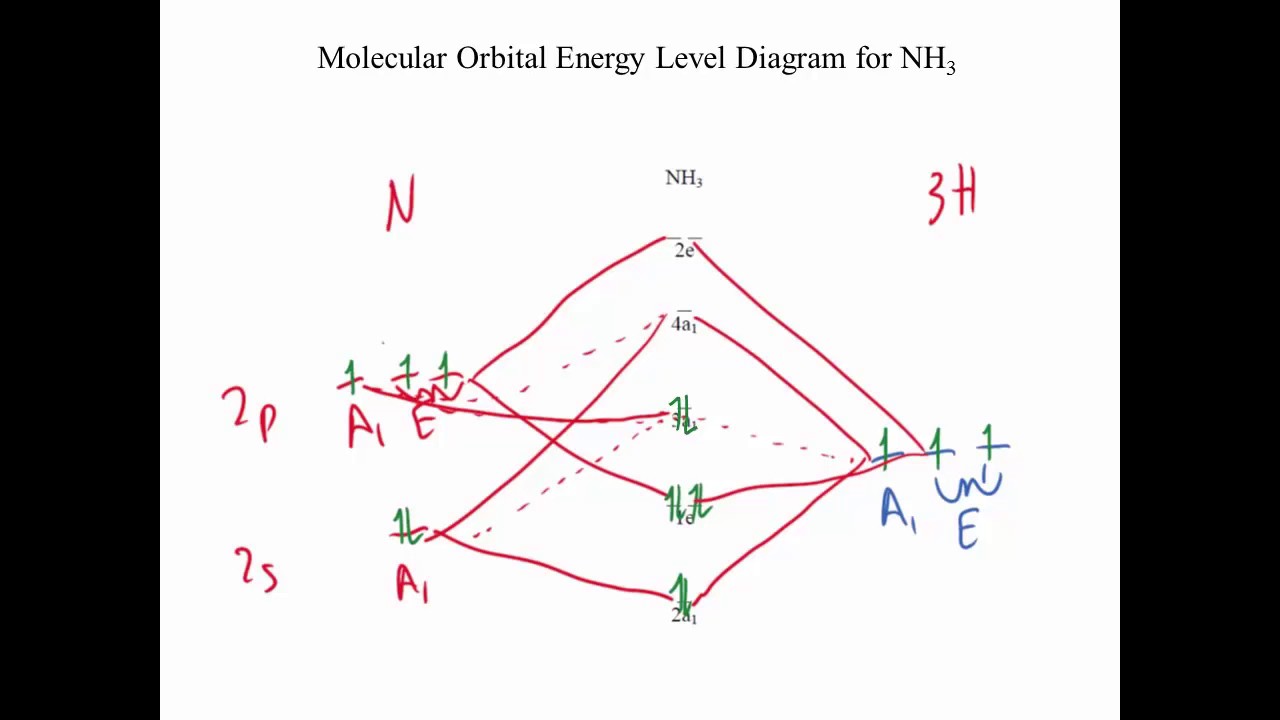
The MO energy diagram for NH3 is shown in Figure 3. The energy.
As can be seen from the energy diagram – four of the molecular orbitals occur as Ammonia has two pairs of degenerate orbitals, one bonding and one.MO Diagram for Triangular H 3 A fragment approach to deriving molecular orbitals Inorganic Chemistry. This lone pair orbital also involves bonding of N 2p z with the bonding MO of the stretched H 3 molecule This MO is responsible for the Lewis base character of the ammonia molecule Inorganic Chemistry.
Specific combinations of atomic orbitals are use to build molecular orbitals. As seen here the p z orbital, like the 1s and 2s orbitals, has A 1 symetry; they are totally symmetric to the operations of the C 3v point group..
The p x and p y are affected by the C 3v point group operations by equal amounts and hence are degenerate. These atomic orbitals have e symmetry as defined by the. The molecular orbital diagram of NH3 is presented in Figure 5 and will be elaborated in regards to its interactions.
The s orbitals for the 3 hydrogens are used to set up the sigma and anti bonding combinations of N sp 3 orbitals and the H 1s orbitals. Molecular Orbital Theory – Walsh diagram The Walsh diagram shows what happens to the molecular orbitals for a set of molecules which are related in structure. In this case, the difference is the H-X-H bond angle which decreases from o to 90 o Molecular Orbital Theory – .
In NH3, the HOMO (Highly Occupied Molecular Orbital) is a mostly nitrogen based orbital that corresponds to the Lone pair of electrons. This is why ammonia acts as a Lewis base at the N atom.
The LUMO (Lowest Unoccupied Molecular Orbital) is the 2e level that has more H character – this shows why NH3 can also act as a Lewis acid through the H 3/5(2).File:Ammonia MO schematron.org – Wikimedia CommonsSALC Ammonia – Molecular Orbitals