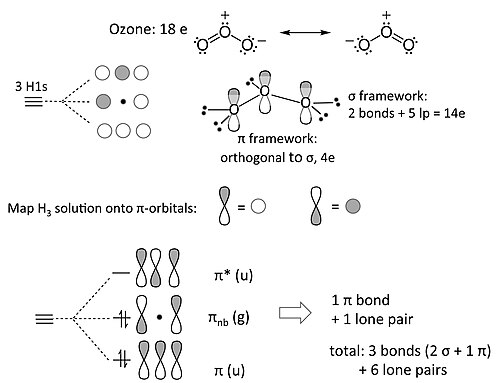
The ozone molecule’s Lewis structure shows that even the preferred structure The pi molecular orbital energy diagram for ozone into which are distributed four . Please draw MO diagram for ozone (O3).
I saw a pic for O3 where the 2s of O was interacting with both bonding and antibonding. Can you please draw and it.
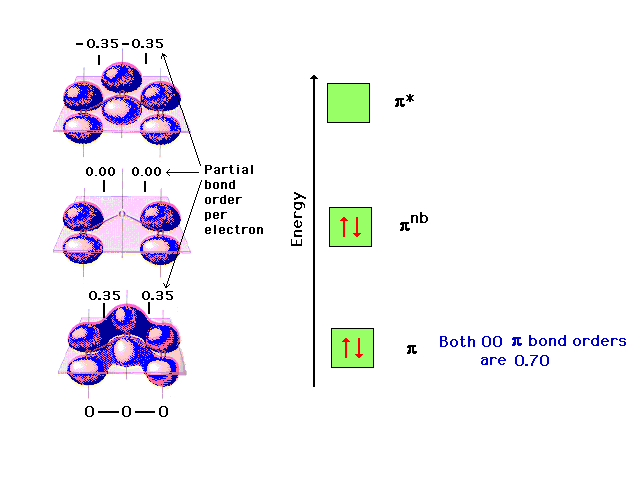
Let’s look at the molecular orbital diagram of ozone. We’ll use the hybrid orbital approximation.
Each oxygen atom combines its 2s, 2pz and 2py orbitals to make . Make sure you understand how the molecular orbital coefficients are determined and their meaning.
MO Diagram CO2
Draw similar diagrams for other orbitals in the print out. Make sure you understand how the molecular orbital coefficients are determined and their meaning.
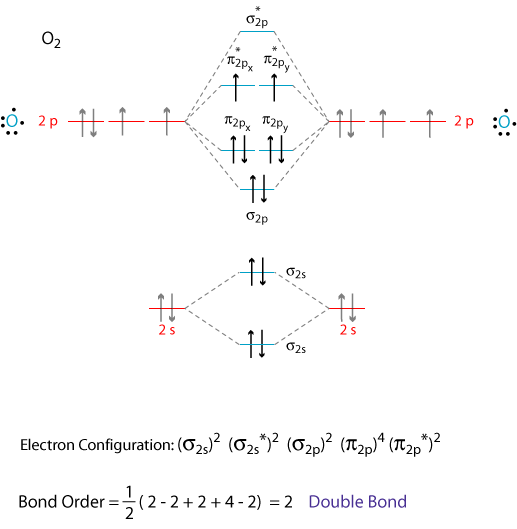
Draw similar diagrams for other orbitals in the print out.The first photo is straight from a edition Pearson general chemistry textbook, and it shows you what the molecular orbital (MO) diagram for O2 is. From that diagram, you can then easily fill out what the O2- and O2+ MO diagrams should be—and that is in the second photo I included.
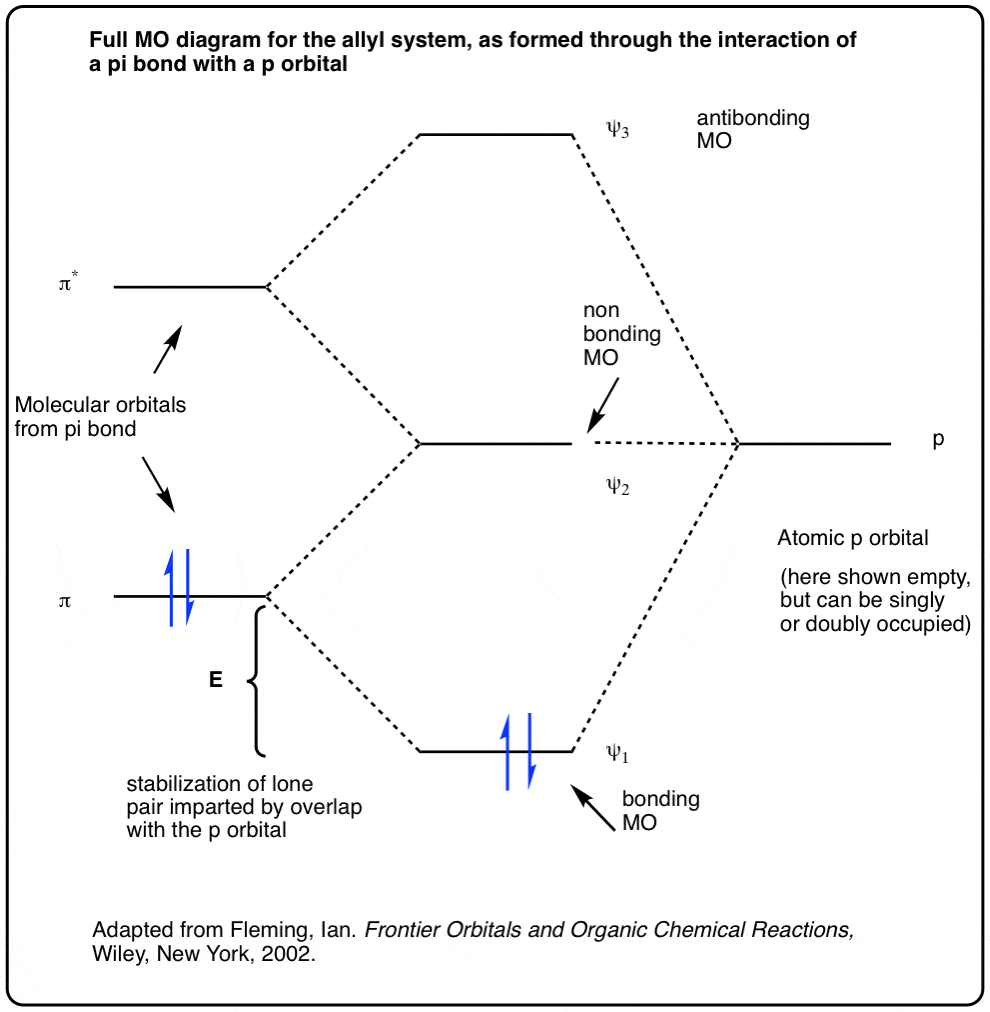
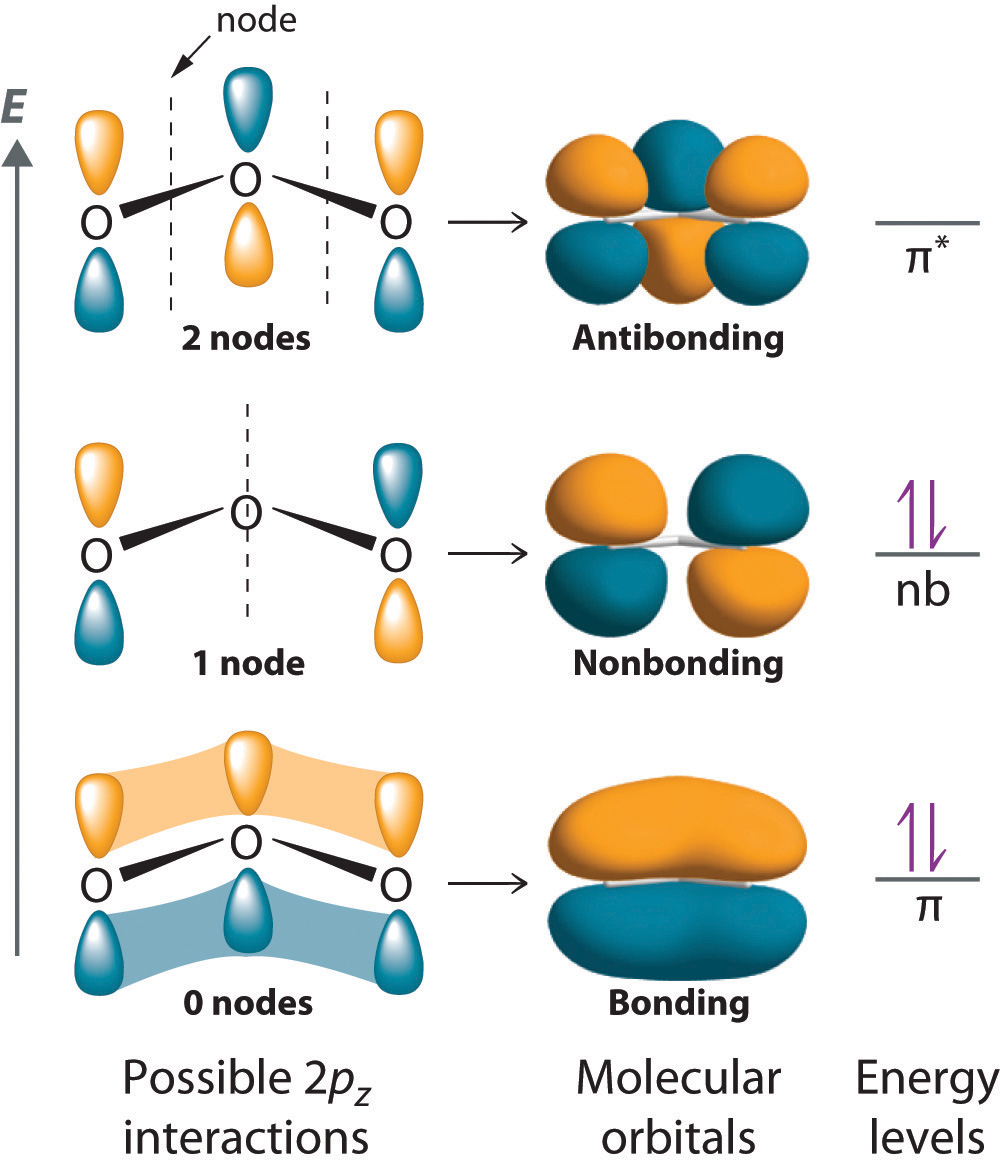
Molecular Orbitals. Delocalized Bonding: Conjugation in Ozone. Ozone is a fairly simple molecule, with only three atoms.
However, in order to focus on one aspect of ozone’s structure, we will use a hybrid approximation in order to simplify the picture. The Lewis structure of ozone is somewhat unsatisfactory.
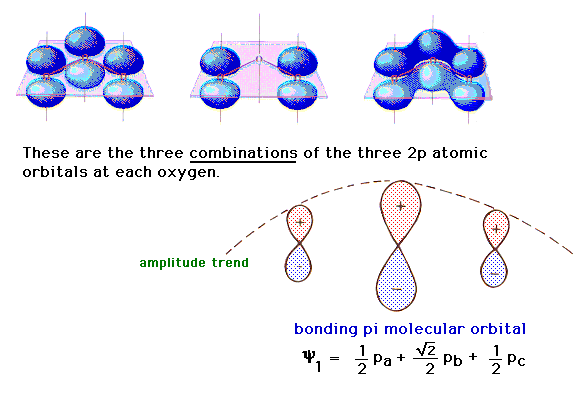
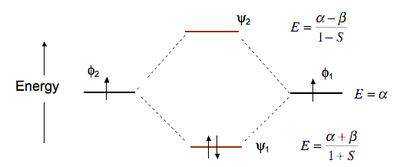
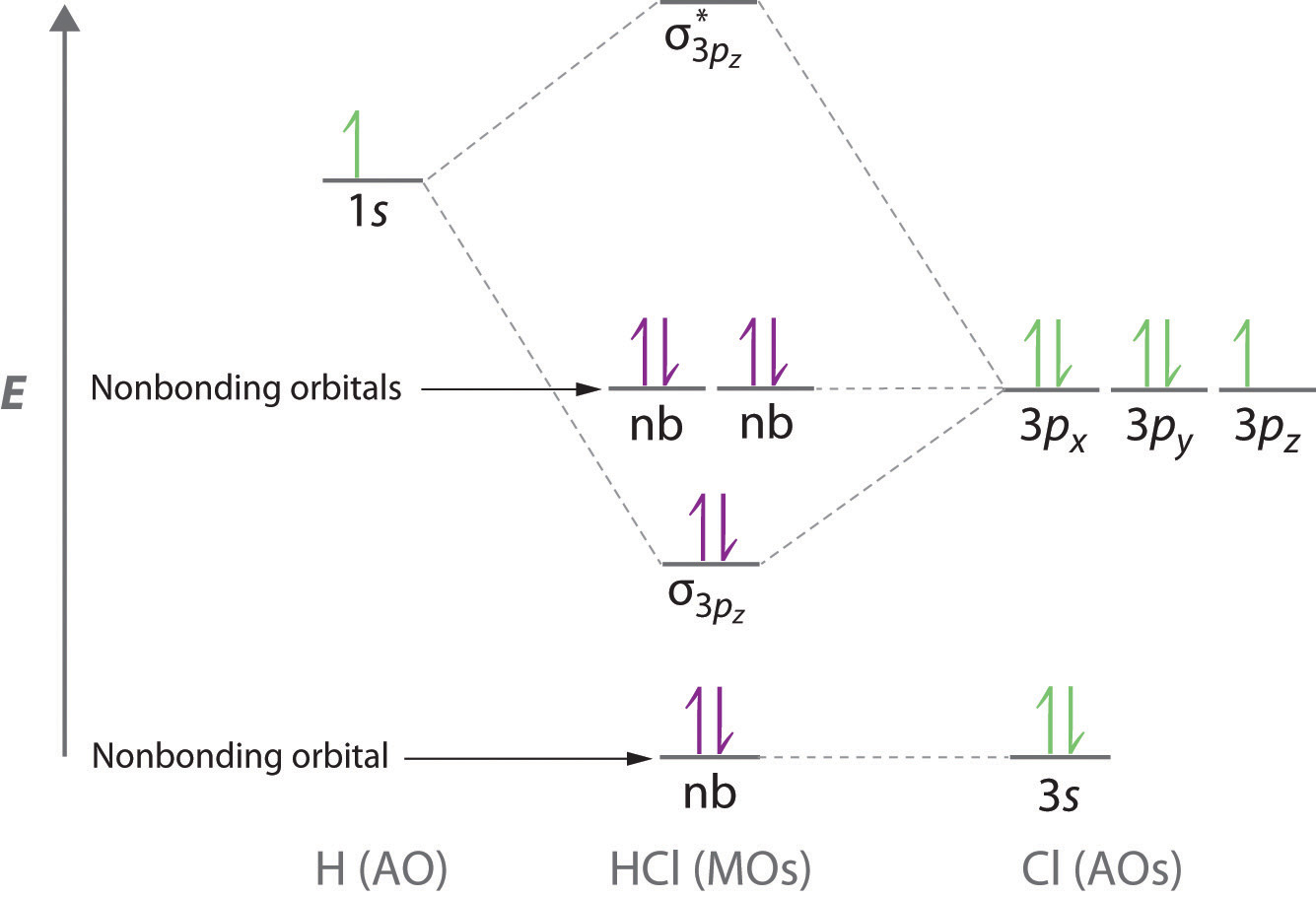
eV. Because of the difference in their atomic orbital energies, the 1s orbital of hydrogen and the 3s orbital of sulfur interact only weakly; this is shown in the diagram by a slight stabilization of the lowest energy molecular orbital with respect to the 3s orbital of sulfur.
This lowest energy orbital is . A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) molecular orbital method in particular.
Compare the bond order in H 2 + and H 2 using the molecular orbital energy diagram for H 2. The bond dissociation energy of the H 2 molecule is kJ/mol.
Explain why .Pi Bonds over 3 AtomsMultiple Bonds in MO Theory – Chemistry LibreTexts