[Bohr Model of Calcium], Number of Energy Levels: 4. First Energy Level: 2.
Bohr Diagrams of Atoms and Ions
Second Energy Level: 8. Third Energy Level: 8.
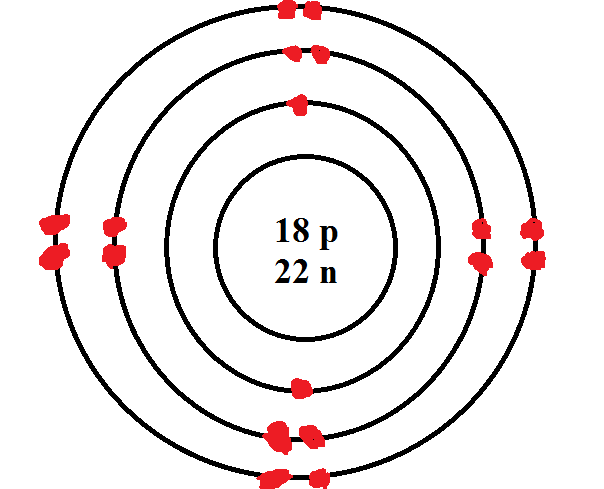
Calcium. This element has 20 protons, 20 electrons, and 20 neutrons giving it an atomic mass of Bohr Model of Calcium. Calcium · Home · More information about Calcium · Fun facts · Calcium Bohr diagram · Citations.
Picture. Powered by Create your own unique website with. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun.
In the Bohr model, electrons are. Calcium.
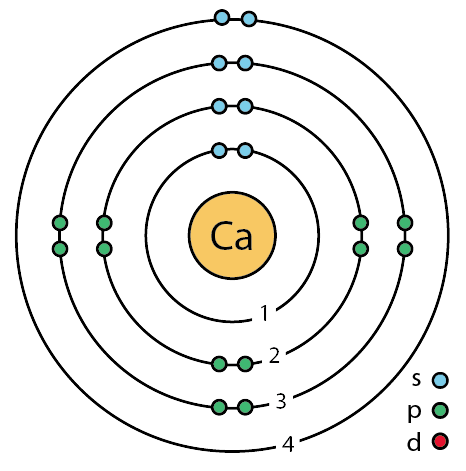
This element has 20 protons, 20 electrons, and 20 neutrons giving it an atomic mass of Bohr Model of Calcium.A Bohr diagram is a simplified visual representation of an atom that was developed by Danish physicist Niels Bohr in The diagram depicts the atom as a positively charged nucleus surrounded by electrons that travel in circular orbits about the nucleus in discrete energy levels. Powered by Create your own unique website with customizable templates. Get Started.
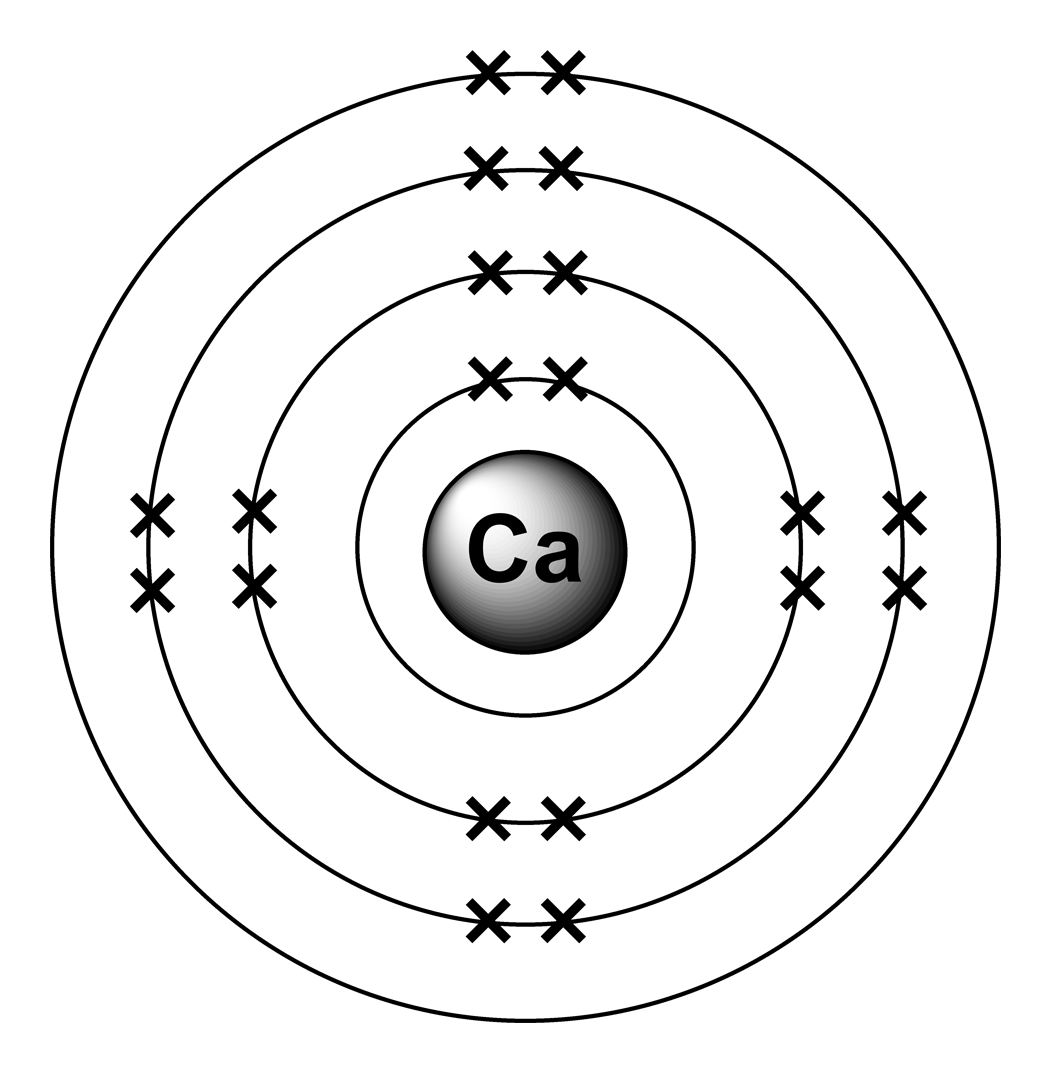
Bohr Diagram For Calcium ~ thanks for visiting our site, this is images about bohr diagram for calcium posted by Alice Ferreira in Bohr category on Nov 06, You can also find other images like wiring diagram, parts diagram, replacement parts, electrical diagram, repair manuals, engine diagram, engine scheme, wiring harness, fuse box, vacuum diagram, timing belt, timing chain, brakes diagram, . A Bohr model of an atom shows a central nucleus which contains protons and neutrons.
The protons have a positive charge, the neutrons are neutral. A model of calcium shows 20 protons.
Most calcium atoms have 20 neutrons (although this can vary). The shells surrounding the nucleus contain the electrons, which have a negative charge. Bohr model is an early model of atomic structure in which electrons circulate around the nucleus in discrete stable orbits with different energy levels.
This model was the first to predict and explain the atomic spectrum of the hydrogen atom, which arises as the electron jumps from one orbit to another orbit of lower energy, giving off electromagnetic radiation of predictable frequencies.Chemical schematron.org – Calcium (Ca)Bohr Rutherford Diagram Of Calcium – schematron.org